Exam 1: Methods of measuring bacterial growth
Estimating the growth of bacteria is extremely important. Environmental health officers regularly inspect food premises and take sample for analysis. Water boards check water supplies daily. Many products are produced using bacteria grown in fermenters. Measuring their growth is an important part of the process.
There are several different methods of measuring growth:
• Rough estimates of growth rates can be made by regularly measuring the diameter of a bacterial or fungal colony as it spreads from a central point to cover the surface of a solid growth medium (such as an agar plate)
• The size of a population of microorganisms in liquid culture may be measured by counting cells directly or by first diluting the original sample and then counting cell numbers (see below), or by taking some indirect method such as the turbidity (cloudiness) of the culture.
Direct cell counts may be divided into:
1. Total counts = which include both living cells and dead cells
2. Viable counts = which count living cells only
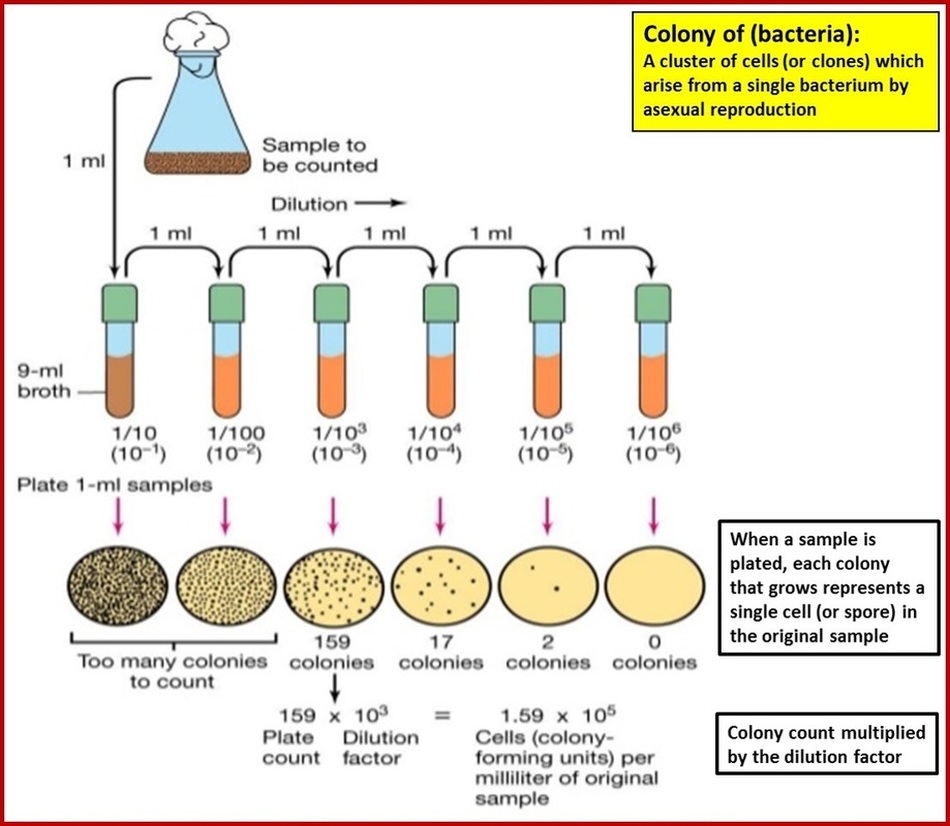
In practice, it is never possible to count whole populations of microorganisms. Instead, the cells in a very small sample of culture are counted, and the result multiplied up to give a population density in organisms per cm3 of culture. Even then, the population density is likely to be so high that cell counts are usually made in known dilutions of the culture, usually in 10-fold steps. This is known as serial dilution.
The dilution plating technique is as follows:
A culture medium, such as milk or a water sample, is made into a series of dilutions using the serial dilution technique.
Estimating the growth of bacteria is extremely important. Environmental health officers regularly inspect food premises and take sample for analysis. Water boards check water supplies daily. Many products are produced using bacteria grown in fermenters. Measuring their growth is an important part of the process.
There are several different methods of measuring growth:
• Rough estimates of growth rates can be made by regularly measuring the diameter of a bacterial or fungal colony as it spreads from a central point to cover the surface of a solid growth medium (such as an agar plate)
• The size of a population of microorganisms in liquid culture may be measured by counting cells directly or by first diluting the original sample and then counting cell numbers (see below), or by taking some indirect method such as the turbidity (cloudiness) of the culture.
Direct cell counts may be divided into:
1. Total counts = which include both living cells and dead cells
2. Viable counts = which count living cells only
In practice, it is never possible to count whole populations of microorganisms. Instead, the cells in a very small sample of culture are counted, and the result multiplied up to give a population density in organisms per cm3 of culture. Even then, the population density is likely to be so high that cell counts are usually made in known dilutions of the culture, usually in 10-fold steps. This is known as serial dilution.
The dilution plating technique is as follows:
A culture medium, such as milk or a water sample, is made into a series of dilutions using the serial dilution technique.
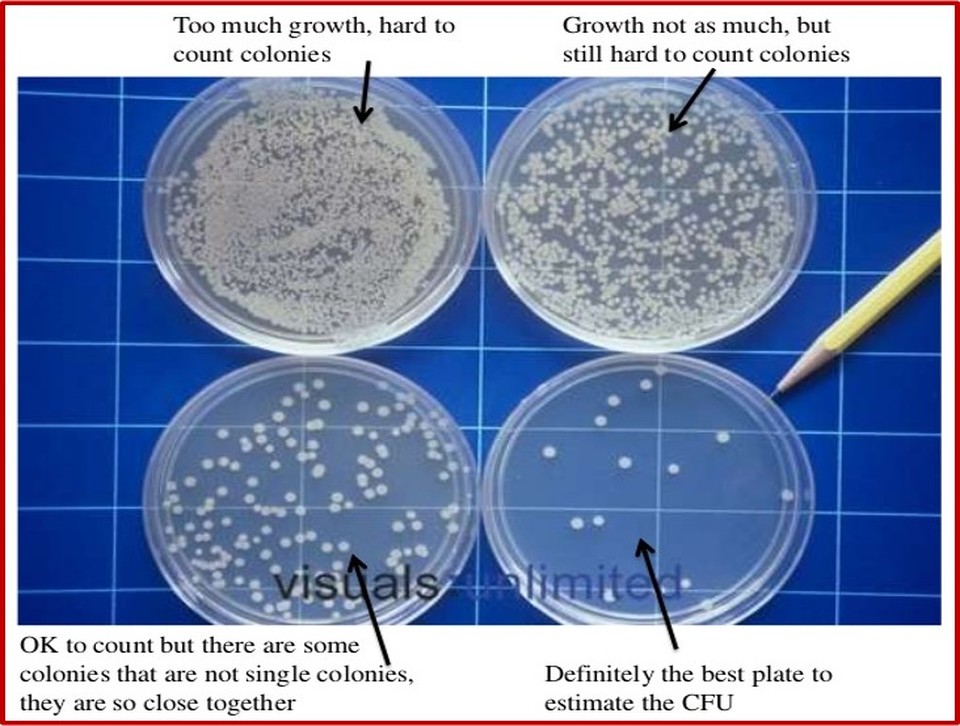
1cm3 (the same as saying 1ml) of each of the diluted samples (see diagram above), are individually streaked onto sterile agar plates, which are then placed in an incubator at 25oC for two days to allow time for the bacteria to grow. After all the streaks have been allowed to grow, the dilution at which the colonies are distinct and separate is noted down (this might be the 1/1000 dilution from the example above). If the dilution is insufficient then colonies will merge, referred to as ‘clumping’, and counting is inaccurate (the example above would be the 1/10 and the 1/100 dilution). The distinct and separate colonies of bacteria from the agar plate you have chosen are counted with the assumption being made that each colony has arisen from a single cell, which has divided asexually, from the original culture medium. To find the total viable cell count the number of colonies you count is multiplied by the appropriate dilution factor. So in the example above this would be:
159 colonies counted on the 1/1000 dilution agar plate
So 159 x 1000 = 1.59 x 10 to the power 5 bacteria in the original sample.
However, if the dilution is insufficient then colonies will merge (see 1/10 and 1/100 dilution agar plates above), referred to as ‘clumping’, and counting is inaccurate, resulting in an underestimate of numbers
159 colonies counted on the 1/1000 dilution agar plate
So 159 x 1000 = 1.59 x 10 to the power 5 bacteria in the original sample.
However, if the dilution is insufficient then colonies will merge (see 1/10 and 1/100 dilution agar plates above), referred to as ‘clumping’, and counting is inaccurate, resulting in an underestimate of numbers
Using a Haemocytometer
A more accurate method involves using a haemocytometer. This is a specialised microscope slide originally used to count red blood cells. Using the haemocytometer gives total cell counts as it is not possible to distinguish between living and dead cells (you are not required to describe or use a haemocytometer.)
Using Turbidimetry
A third method, known as turbidimetry, involves using a colorimeter to measure the cloudiness or turbidity of the culture as cell numbers increase. Results are derived by comparison with a standard graph of light absorbance plotted against known cell numbers (You are not required to describe or use a calorimeter).